- Published on
What happens to CCS captured carbon
- Authors
-
-

- Name
- Vivienne Roberts
- in/viviroberts
-
Geological storage:
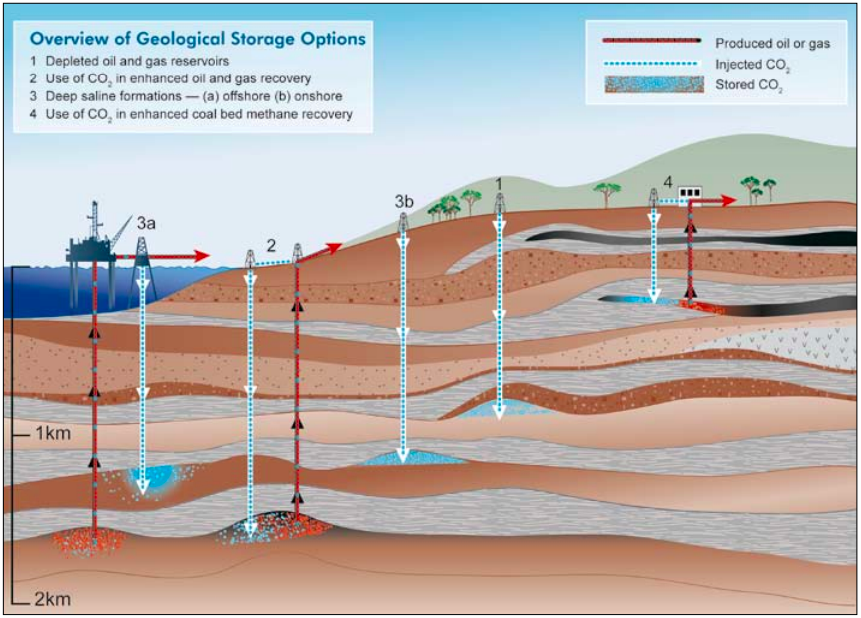
Carbon Capture and Storage or Sequestration typically conjures images of the injection of carbon into a suitable underground cavity, with appropriate geological characteristics. The CO2 captured is then permanently trapped within these cavities. As shown in the diagram below, there are a number of types of cavities or geological structures that can be used.
Source: IPCC Special report on SRCCS
Options 2 and 4 above make use of CO2 as an input in an industrial process; namely to assist with the recovery of oil, gas or coal bed methane. CO2 is injected and the pressure assists in forcing more fuel upwards. Effectively this is storing CO2 to assist with accessing more fossil fuels which will result in more CO2. However, the use of CO2 helps to decrease the net emissions in the overall process, based on the assumption that the oil would have been accessed in the first place. If pressurised CO2 is injected into the cavity after the completed depletion of the reservoir, it could also be considered to be a net sink.
Option 3 involves the injection of CO2 into a saline, or brine, solution. This is water that is rich in minerals, and unsuitable for agricultural purposes. This may not be suitable for areas aiming to access geothermal potential of underground water deposits.
Mineral carbonation:
This involves the formation of stable carbonate materials through controlled chemical reactions with calcium or magnesium compounds. The output of this process can be used in construction or building projects, and the carbon is permanently trapped. There are naturally challenges associated with this process, not least of which is the mining required for the minerals to be used as inputs to the process. The capture of carbon and the chemical process itself requires energy. The IPCC considers that such a facility would require 60-180% more energy than a power facility without carbon capture or mineral carbonation processes.
SOURCE: IPCC SPECIAL REPORT ON SRCCS
Industrial applications:
CO2 is used as an input in a number of industrial processes. The potential for diverting CO2 captured and using this in such processes is being explored. One of the key considerations relates to the resulting industrial output, and whether the CO2 remains permanently stored therein.
Excluding CO2 as an input in enhanced oil recovery (discussed above), CO2 is also used as a reagent in fertiliser manufacture, methanol production or is used for other applications, such as a refrigerant, in fire extinguishers, in soft drink carbonisation, in plastics production or in algae cultivation. As the IPCC indicates in their special report on CCS (link above),_ “as a measure for mitigating climate change, this option is meaningful only if the quantity and duration of CO2 stored are significant, and if there is a real net reduction of CO2 emissions. The typical lifetime of most of the CO2 currently used by industrial processes has storage times of only days to months. The stored carbon is then degraded to CO2 and again emitted to the atmosphere. Such short time scales do not contribute meaningfully to climate change mitigation.” _
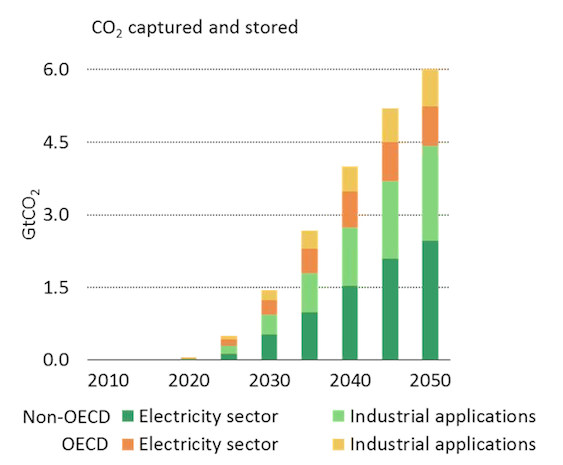
Given that nearly half of all carbon capture and storage is expected to be within the industrial sector (including EOR), it is important that the effectiveness of the solutions are considered and monitored. Particularly as the IPCC states that, “in view of the low fraction of CO2 retained, the small volumes used and the possibility that substitution may lead to increases in CO2 emissions, it can be concluded that the contribution of industrial uses of captured CO2 to climate change mitigation is expected to be small.”
[Source: IEA Energy Technology Perspectives 2015 presentation]
Where this becomes important is in the cost of the overall CCS system. Where CO2 is considered to be an input into an industrial process, the cost of separating carbon at a power station can be offset against its value in the process. Storage for the sake of storage (e.g. underground storage without EOR applications) cannot be offset, unless other energy generation options are suitably priced for carbon.